A WORD FROM OUR CHIEF SCIENTIFIC OFFICER
Dear FAPI Community,
Happy 2026!
Since last connecting through this newsletter, SOFIE is pleased to share that our Phase 3 [18F]FAPI-74 diagnostic studies, FAPI-GO (FAPI in Gastroesophageal Oncology, NCT07217704) and FAPI-PRO (FAPI in Precision Imaging of Pancreatic Cancer, NCT07217717) are both activated with patient recruitment well underway. The clinical trials are planned for 18 sites with an estimated enrollment of 200 subjects over a 24-month period.
I was honored to participate in the 8th Theranostics World Congress held in Cape Town, South Africa, earlier this year. FAP targeting was a hot topic and I was buoyed by all the engagement about its future directions. Members of our FAPI team will be attending a slate of scientific meetings this spring. Please see details below – we would love to see you in person!
We are excited to kick off another year with a focus on further educating the community about the potential of the FAP biomarker. Let us know what you’d like to know more about!
– Sherly Mosessian, Ph.D., Chief Scientific Officer

[18F]FAPI-74 CLINICAL DEVELOPMENT UPDATE
FAPI ON THE MOVE
A member of SOFIE’s FAPI team will attend the spring meetings listed below. If you are attending, we would love to meet you and share more about our FAPI program.
For the latest FAPI updates, visit SOFIE’s LinkedIn page!
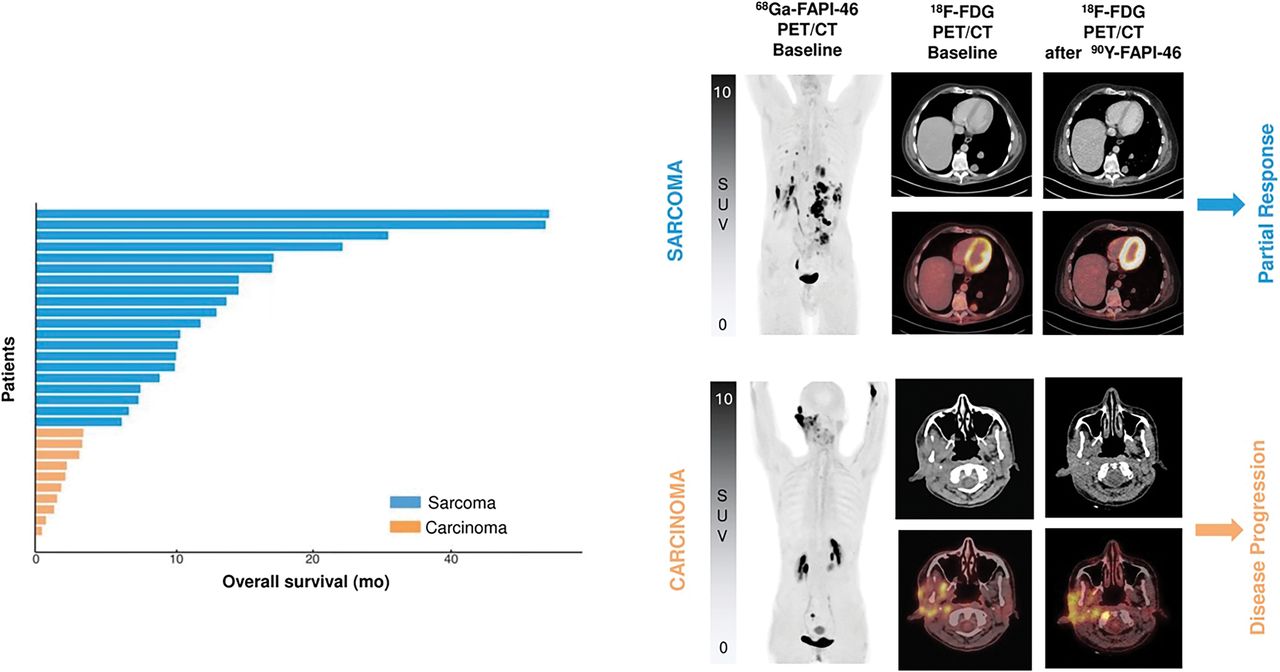
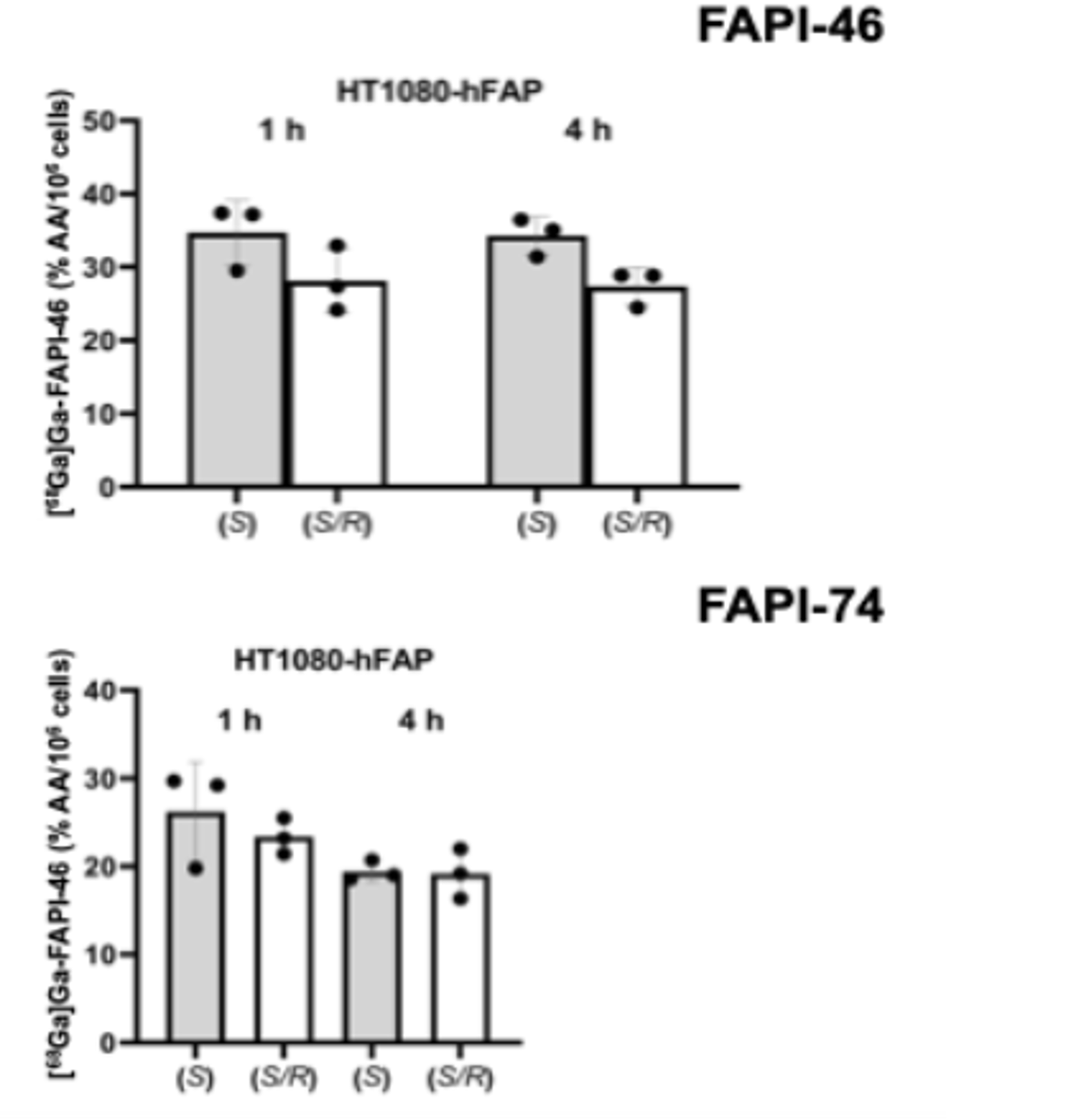
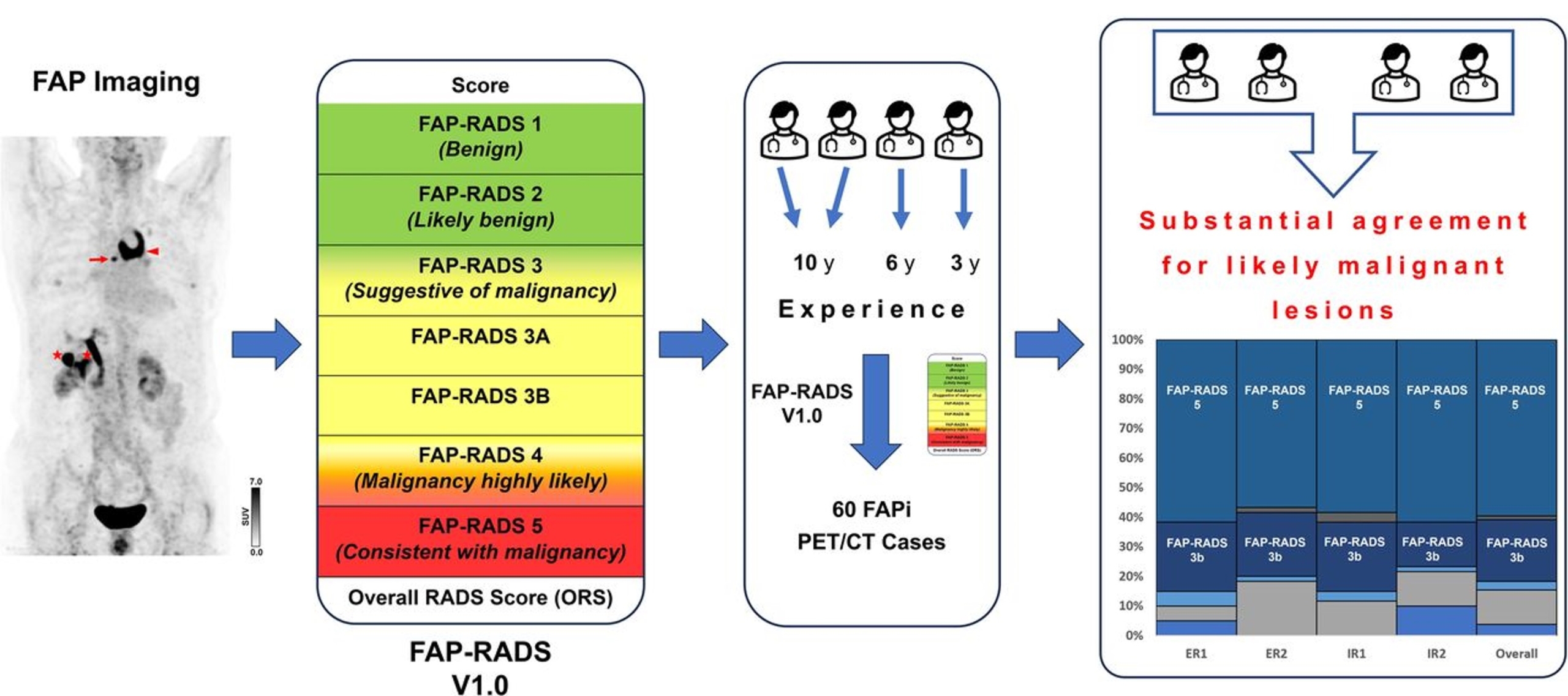
PUBLICATION HIGHLIGHTS
Each newsletter will feature recent FAPI publications.
FAPI OUTREACH PROGRAM
Join us in advancing the frontier of molecular imaging through the FAPI Outreach Program
SOFIE is continuing to accept applications for our FAPI Outreach Program. This program aims to raise awareness and foster collaboration in the field of molecular imaging, specifically focusing on Fibroblast Activation Protein Inhibitor (FAPI) studies. This program seeks to engage with healthcare professionals, researchers, and the broader community to share knowledge, advancements, and the potential impact of FAPI in diagnostic and therapeutic applications.
*GE Healthcare licensed products and regions are accepting new applications.