Emil Novruzov, Eduards Mamlins, Liam Widjaja, Sophie C Siegmund, Tadashi Watabe, Yuriko Mori, Christian Stief, Amanda Tufman, Ken Herrmann, Michael A Gorin, Martin G Pomper, Steven P Rowe, Frederik L Giesel, Rudolf A Werner, Gabriel T Sheikh
Abstract:
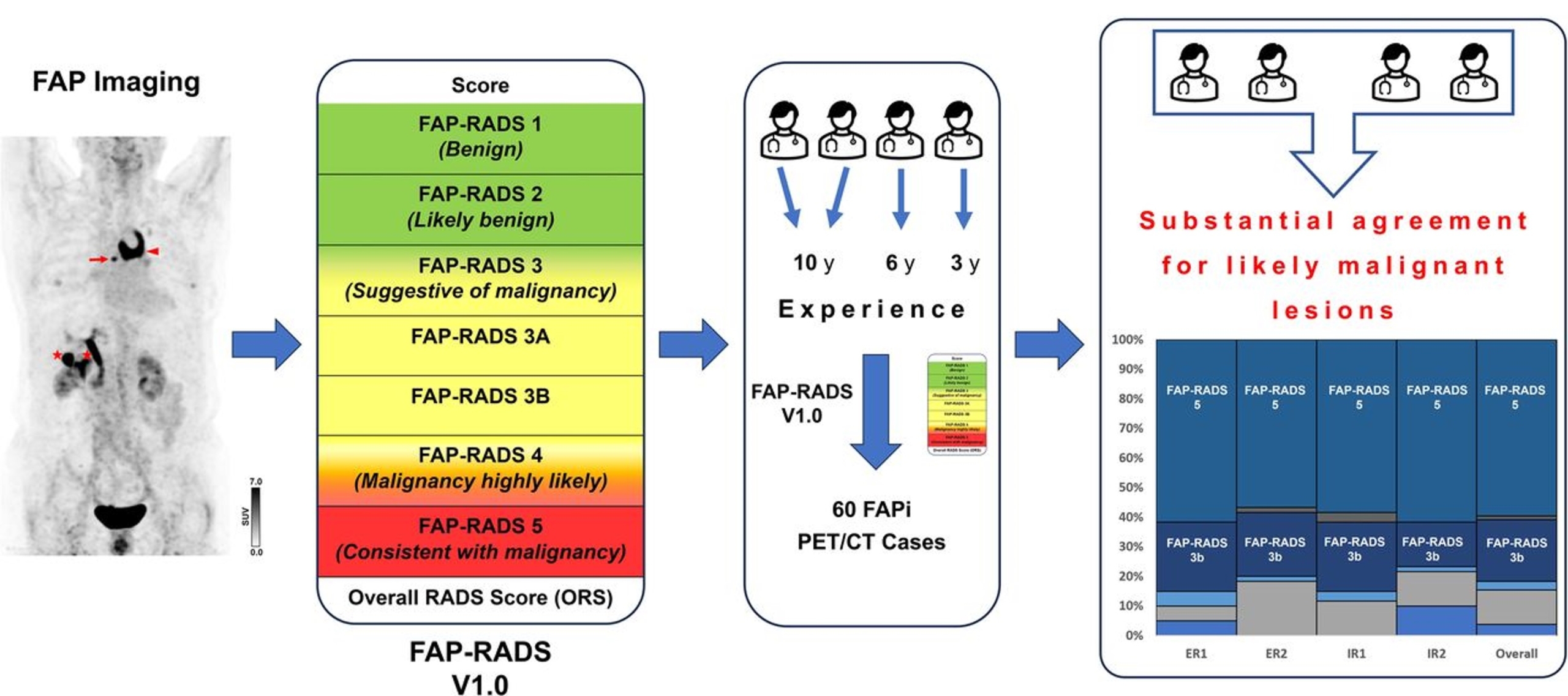
Recently, a collaborative effort among research centers in the United States and Europe resulted in the development of fibroblast activation protein reporting and data system (FAP-RADS) version 1.0, a reporting framework for fibroblast activation protein (FAP)-targeted imaging using the fundamental principles of the molecular imaging reporting and data systems framework. This retrospective study aimed to investigate the reliability and consistency of FAP-RADS version 1.0 framework for patients with lung cancer and urothelial cancer.
Methods: Four readers with varying levels of PET/CT experience, working without access to clinical patient information (2 readers with more than 10 y of experience, 1 reader with 6 y of experience, and a nuclear medicine resident with less than 3 y of experience) evaluated PET/CT scans from a total of 60 patients (30 with lung cancer and 30 with urothelial cancer). Readers were instructed to identify no more than 5 target lesions (TLs) per patient, with a maximum of 3 TLs per organ or compartment. Each evaluation required the assignment of a final overall FAP-RADS score, defined as the highest score among the assessed TLs.
Results: Interreader agreement for the overall scan impression and the presence of lymph node metastases was moderate to substantial, with κ values of 0.62 (95% CI, 0.52-0.73) and 0.59 (95% CI, 0.49-0.69), respectively. Interreader agreement regarding referral for radiopharmaceutical therapy on the basis of tracer uptake intensity and disease extent were high, with κ values of 0.70 (95% CI, 0.60-0.81) and 0.66 (95% CI, 0.55-0.76), respectively. The internal consistency for TL-based assessments and overall FAP-RADS score was almost perfect, with a Cronbach α of 0.92 and 0.88, respectively. Analyses of TL-based FAP-RADS scores demonstrated high interreader agreement for both experienced and less experienced readers, with weighted κ values of 0.72 (95% CI, 0.53-0.91) and 0.75 (95% CI, 0.57-0.92), respectively.
Conclusion: The FAP-RADS version 1.0 framework appears to be a robust tool for standardized reporting in both clinical studies and routine clinical practice, demonstrating substantial interreader agreement across most evaluated categories. Further studies are warranted to validate its applicability in additional tumor entities and with other FAP-targeting ligands.

![FAP Expression in Renal Tumors Assessed by [68Ga]Ga-FAPI-46 PET Imaging and FAP Immunohistochemistry: A Case Series of Six Patients](https://sofie.com/wp-content/uploads/2025/12/info.ibamolecular-scaled-500x383.jpg)