Magdalena Staniszewska, Ralph Hübner, Camilla Locatelli, Douglas Howard, Matilde Forni, Francis Man, Bent Wilhelm Schoultz, Syed Nuruddin, Ingrid Sofie Norberg-Schulz Hagen, Sherly Mosessian, Ken Herrmann, Valeska von Kiedrowski, Katharina Lückerath
Abstract:
Background: Enantiomeric composition may impact binding and potentially performance of a drug product but is not routinely tested and minimum acceptance criteria are not always established. Some batches of the precursors FAPI-46 and FAPI-74 used clinically contain a mixture of (S)- and (R)-enantiomers. This study investigates the in vitro pharmacodynamic characteristics of different enantiomeric compositions of FAP-targeting radioligands FAPI-46 and FAPI-74 and in vivo pharmacokinetics of FAPI-46 in naïve mice.
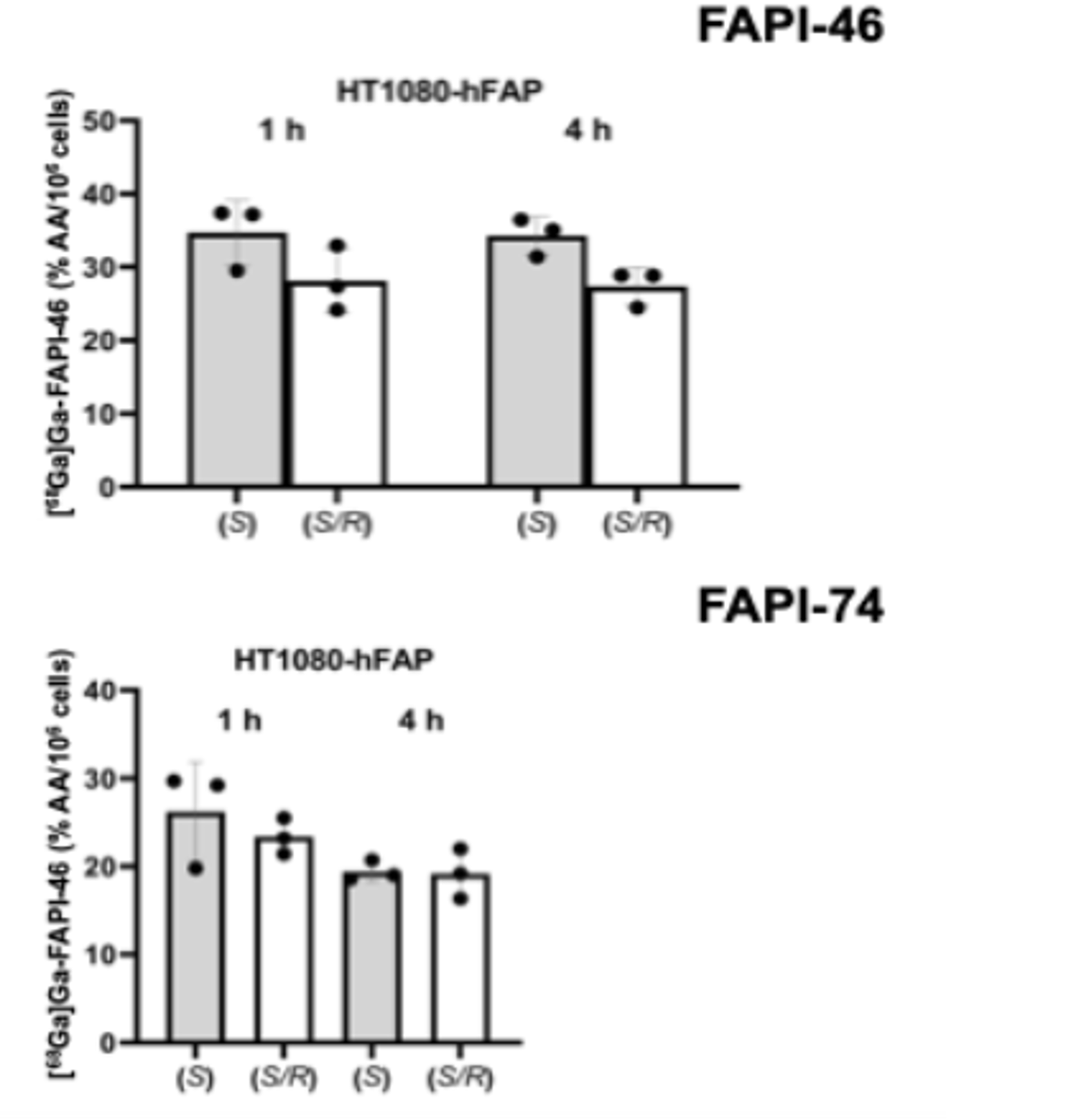
Results: (S)-, (R)-, and (S/R)-enantiomers of radiolabeled FAPI-46 and FAPI-74 were evaluated for stability in human serum, binding affinity, internalization, retention, and selectivity using HT1080-hFAP cells and HEK293-hCD26 cells. All enantiomeric compositions showed high stability in human serum with minimal serum protein adhesion. (S)- and (S/R)-enantiomers exhibited comparable binding, internalization, and retention characteristics. High selectivity for FAP over CD26/DPP4 was maintained. (R)-enantiomers showed no specific binding to FAP. Additionally, the same enantiomers of 68Ga-labeled FAPI-46 were evaluated by dynamic positron emission tomography (PET) imaging (0-60 min post-injection) and ex vivo organ biodistribution in naïve BALB/c mice (10, 30, and 60 min, or 4 h post-injection) to compare their pharmacokinetic profiles. PET imaging revealed nearly identical time-activity curves for the enantiomers of [68Ga] Ga-FAPI-46, indicating similar whole-body distribution within 1 h post-injection, with rapid renal clearance and minimal muscle uptake. Ex vivo organ biodistribution confirmed comparable pharmacokinetic profiles between enantiomers, with no significant differences in tissue distribution.
Conclusion: These findings provide important validation that current clinical (S/R)-mixtures of FAPI-46/-74 exhibit pharmacokinetic and pharmacodynamic behavior comparable to pure (S)-enantiomers, supporting regulatory confidence and cross-trial reproducibility in the global FAPI-46/-74 imaging landscape.

![FAP Expression in Renal Tumors Assessed by [68Ga]Ga-FAPI-46 PET Imaging and FAP Immunohistochemistry: A Case Series of Six Patients](https://sofie.com/wp-content/uploads/2025/12/info.ibamolecular-scaled-500x383.jpg)