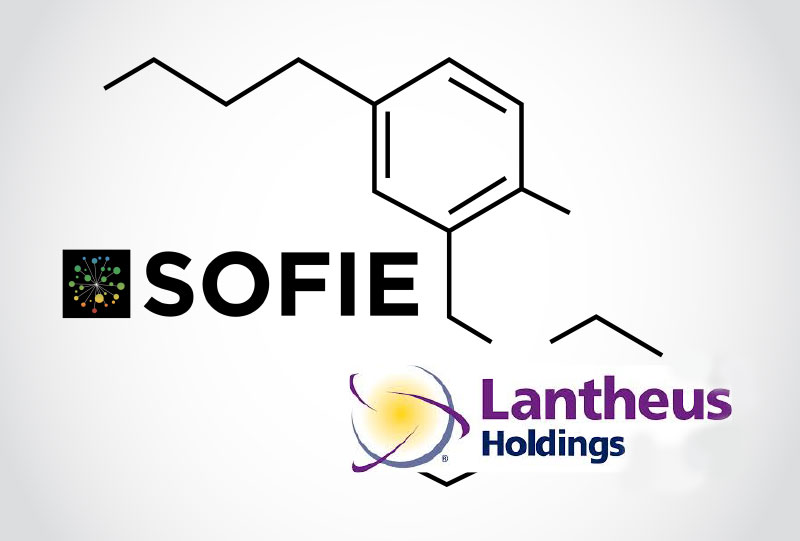
SOFIE, an established Radiopharmaceutical Contract Manufacturing Organization and GMP radiopharmacy network, has signed a commercial supply agreement with Lantheus Holdings, Inc. (the parent company of Progenics Pharmaceuticals, Inc. (Progenics)) to manufacture and distribute Lantheus’ lead investigational prostate cancer imaging agent, PyL™ (18F-DCFPYL).
Following U.S. Food and Drug Administration (“FDA”) approval, Lantheus will have a critical need for dependable and scalable manufacturing and distribution partners to support PyL™. Under the commercial supply agreement, SOFIE will roll out PyL™ via its national network, which has manufactured PyL™ for Progenics during the clinical trials. SOFIE’s network will help meet the expected commercial demand for PyL™ by leveraging its expertise, experience, and distribution capabilities to achieve widespread availability and market coverage.
SOFIE’s CEO, Patrick Phelps stated, “Our relationship with Progenics, and now Lantheus, as well as our network capabilities have been greatly refined over the years we’ve worked on PyL™. With over 500 doses previously manufactured for Progenics’ clinical trials, SOFIE is committed to delivering the doses needed, for widespread use post-FDA approval.” SOFIE’s Chief Technical Officer, Dr. Melissa Moore, added, “Solidifying our relationship with Lantheus via this commercial agreement is the next step in the evolution of our Radiopharmaceutical Contract Manufacturing Organization. SOFIE believes that PyL™ will be an important next generation prostate cancer diagnostic tool and will offer new opportunities to those suffering from this common form of cancer.”
Paul Blanchfield, Lantheus Chief Commercial Officer commented, “We are delighted to partner with SOFIE as we work toward FDA approval to bring PyL™ to the U.S. prostate cancer community. Consistent, reliable, and on-time delivery of diagnostic PET imaging agents is critical to meeting our supply demands and we look forward to our continued partnership with SOFIE to help patients gain access to PyL™.”
About SOFIE
SOFIE’s vision is to improve patient outcomes by developing and delivering molecular diagnostics and therapeutics (Theranostics). With its robust radiopharmaceutical production and distribution network, mature contract manufacturing services, and now, high value Theranostic intellectual property, SOFIE is poised to deliver on the promise of nuclear medicine. For more information, please visit https://sofie.com or contact info@sofie.com
About PyL™ for PET Imaging of Prostate Cancer
PyL™ (also known as 18F-DCFPyL) is a fluorinated prostate specific membrane antigen (PSMA)-targeted positron emission tomography (PET) imaging agent that enables visualization of both bone and soft tissue metastases to determine the presence or absence of recurrent and/or metastatic prostate cancer.
About Prostate Cancer
Prostate cancer is the second most common form of cancer affecting men in the United States: an estimated one in nine men will be diagnosed with prostate cancer in his lifetime. The American Cancer Society estimates that each year approximately 191,930 new cases of prostate cancer will be diagnosed and about 33,330 men will die of the disease. Approximately 3.1 million men in the U.S. currently count themselves among prostate cancer survivors.

![SOFIE Biosciences Announces First Patient Dosed in [18F]FAPI-74 Phase 3 Study for Pancreatic Cancer](https://sofie.com/wp-content/uploads/2026/02/FAPIPRO_SOFIE2-500x383.jpg)
![SOFIE drives U.S. manufacture of [18F]FAPI-74](https://sofie.com/wp-content/uploads/2026/01/FAPI-Site-Map-2026-updated-500x383.png)

![SOFIE Biosciences Announces First Patient Dosed in [18F]FAPI-74 Phase 3 Study](https://sofie.com/wp-content/uploads/2025/12/P4_SOFIE_image-for-news-post-on-website-500x383.jpg)
![SOFIE activates FAPI-PRO, second [18F]FAPI-74 Phase 3 trial](https://sofie.com/wp-content/uploads/2025/12/FAPI_SOFIE-500x383.jpg)