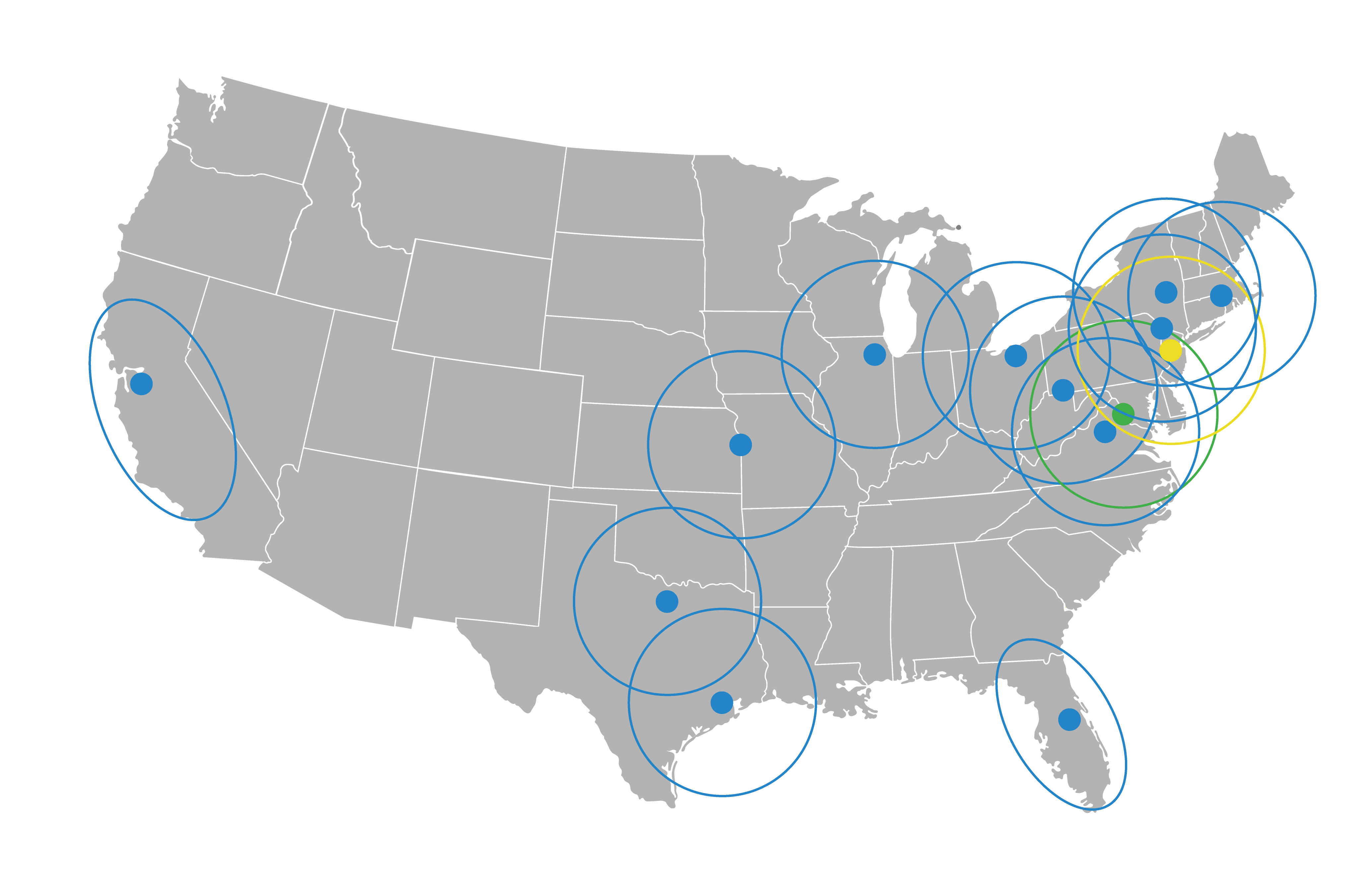
NETWORK
RADIOPHARMACIES
SOFIE has built a high-capacity PET radiopharmaceutical network capable of delivering large volumes of products to as many patients as possible. Our product lines include FDG, Pylarify, Neuraceq, and other clinical trial products.
CAPABILITIES
0
cyclotrons
0+
doses per year
0
dispensing hot cells
0
staff in network
WHAT’S TO COME
We are passionate about PET and believe there will be tremendous growth and exciting new products in the coming years. SOFIE has committed to reinvesting in our business to support the growth of this exciting industry. We recently added two new cyclotrons to the network and will continue this pace through next year and beyond. Each of our sites will have dual cyclotrons and dual dispensing hot cells to support the high volume and reliability necessary for our customers and patients.